Alzheimer’s drugs hold promise for autism
A growing number of studies suggest connections between Alzheimer’s disease, fragile X syndrome and autism, which could point the way to potential treatments.
-
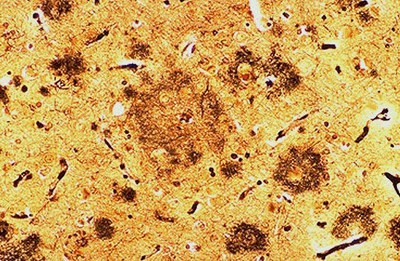
Brain damage: Postmortem brains from some people with autism have elevated levels of APP-beta, the peptide that forms plaques in Alzheimer’s brains.
-
Brain damage: Postmortem brains from some people with autism have elevated levels of APP-beta, the peptide that forms plaques in Alzheimer’s brains.
Fragile X syndrome and autism are developmental disorders that take root in the brain as it forms connections early in life. Alzheimer’s disease, in contrast, is degenerative and progresses as people age. But a number of studies are showing connections between these three disorders. And this may point the way to potential treatments, researchers say.
The strongest link between the disorders is through amyloid precursor protein (APP) — the infamous protein that contributes to plaques in Alzheimer’s brains. Studies have found elevated levels of APP in the blood and brains of individuals with fragile X syndrome or autism and in mouse models of fragile X syndrome1, 2, 3.
This connection is strong enough for some researchers to suggest testing Alzheimer’s drugs, which typically target APP and its byproducts, in autism and fragile X syndrome.
“We really have very little to offer [people with fragile X syndrome],” says James Malter, chair of pathology at University of Texas, Southwestern in Dallas. “Figuring out what APP is doing in the brain is a challenge, but it leads to a potential therapy and we really need to take a leap of faith on this.”
Other researchers caution that without knowing more about the mechanism behind the pathway, this approach is premature and potentially dangerous.
Pharmaceutical companies have spent billions of dollars developing and approving drugs for Alzheimer’s disease. However, most of them show only minor benefit, and a few have even led to fatal side effects in clinical trials.
Treatment target:
Malter and his collaborator Cara Westmark discovered a link between fragile X syndrome and APP serendipitously in 2007. The protein missing in fragile X syndrome, called FMRP, keeps the production of other proteins in check. The researchers thought to use APP as an example of a protein that FMRP does not regulate.
To their surprise, they found that APP is one of FMRP’s targets. They later confirmed that APP levels are elevated in fragile X mouse models, which lack FMRP1.
In a 2011 study, the team showed that lowering APP levels in fragile X mice alleviates several fragile X symptoms, including severe seizures2. This suggests that elevated APP may be responsible for at least some of the symptoms of fragile X syndrome, says Westmark, senior scientist at the Waisman Center at the University of Wisconsin-Madison.
Other researchers caution that this is just the beginning of the story, however.
“These papers show some interesting possible links, but the mechanism is not very well worked out so far,” says Raymond Kelleher, assistant professor of neurology at Massachusetts General Hospital in Boston, who was not involved in the study.
Researchers have been studying the links between APP and Alzheimer’s disease for decades, but its role is still unclear.
Two enzymes cut the APP protein into two mutually exclusive short fragments, or peptides. One of these, APP-beta, forms large plaques in the brains of people with Alzheimer’s disease. Many researchers believe that these clumps are what cause the brain to degenerate.
Some studies suggest that APP-beta may also be elevated in the brains of people with fragile X syndrome and autism.
Westmark and Malter found more APP-beta in the postmortem brains of four people with fragile X syndrome than in control brains, although these results are not statistically significant. And a study published last year in PLoS One found more APP-beta in 20 postmortem brains from people with autism than in those from 8 controls3.
Brain booster:
Malter says he is eager to test drugs that lower APP-beta levels in people with fragile X syndrome. But that may mean boosting levels of APP-alpha instead. Some studies have also suggested that APP-alpha may be linked to autism symptoms.
For example, a 2011 study in PLoS One found more APP-alpha and less APP-beta in the blood of 15 children with severe autism than in 18 controls or 6 children with more mild autism4.
APP-alpha helps build neuronal connections, which makes sense in light of studies that suggest excess neurons and abnormally strong local brain connections in people with autism, says Debomoy Lahiri, professor of psychiatry at Indiana University in Indianapolis and lead investigator of the PLoS One study.
Malter agrees that it is possible that APP-alpha, rather than APP-beta, is the culprit in autism and fragile X syndrome.
“That’s what’s so complex about APP biology,” he says. “We’re looking at the effects of multiple fragments, all of which are biologically active and all of which are present at the same time. It’s very hard to tease apart one from the other.”
One caveat of blood-based studies is that low levels of a peptide in the blood may be an indication that it’s accumulating in the brain, says Malter. This would once again support a role for APP-beta in autism, suggesting that people with the disorder have low blood levels of APP-beta in the blood and high levels in the brain.
Preliminary research shows that one Alzheimer’s drug, memantine, which lowers overall APP levels5, may prove helpful in autism.
Autism-like behaviors in mice lacking one copy of the autism-linked gene MEF2C improve when treated with memantine, according to unpublished results presented in January at the Salk Institute, Fondation IPSEN and Nature Symposium on Biological Complexity in La Jolla, California.
Memantine may also enhance the efficacy of risperidone in children with autism. Risperidone is an antipsychotic and one of only two drugs approved by the U.S. Food and Drug Administration to treat autism6.
By the same token, therapies developed for fragile X syndrome and autism may be beneficial for people with Alzheimer’s disease, says Sean McBride, psychiatry resident at the University of Pennsylvania.
In particular, one promising class of fragile X treatment that targets a receptor called mGluR5 may influence APP levels.
“Counterintuitively, the Alzheimer’s disease field could actually gain more by the movement that has been made in the fragile X field,” he says.
References:
1: Westmark C.J. and J.S. Malter PLoS Biol. 5, e52 (2007) PubMed
2: Westmark C.J. et al. PLoS One 6, e26549 (2011) PubMed
3: Wegiel J. et al. PLoS One 7, e35414 (2012) PubMed
4: Ray B. et al. PLoS One 6, e20405 (2011) PubMed
5: Ray B. et al. Neurosci. Lett. 470, 1-5 (2010) PubMed
6: Ghaleiha A. et al. Int. J. Neuropsychopharmacol. Epub ahead of print (2012) PubMed
Explore more from The Transmitter

Crowdsourcing to curb aggression in autism: Q&A with Matthew Goodwin

Brain connectivity and letting the data speak with Emily Finn
