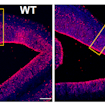
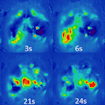
On January 26, 2018, SFARI held a workshop to discuss the use of zebrafish in autism research. Experts in both rodent and fish models discussed key issues in the field, including zebrafish mutant construct validity, the visualization of circuit development and function, and high-throughput phenotyping and pharmacological screens for translational opportunities.